Patterning and Signaling Pathways in the Embryonic Forebrain
A critical aspect of central nervous system development is the production of specialized cell types in the correct locations at the right times in development. One solution to this challenge is to establish spatially segregated germinal zones dedicated to produce specific cell types. This spatial patterning is largely achieved by morphogen signaling during early CNS development. Additionally, progenitor cells in these germinal zones generate multiple cell types in temporally regulated phases, thus achieving both spatial and temporal specificity.
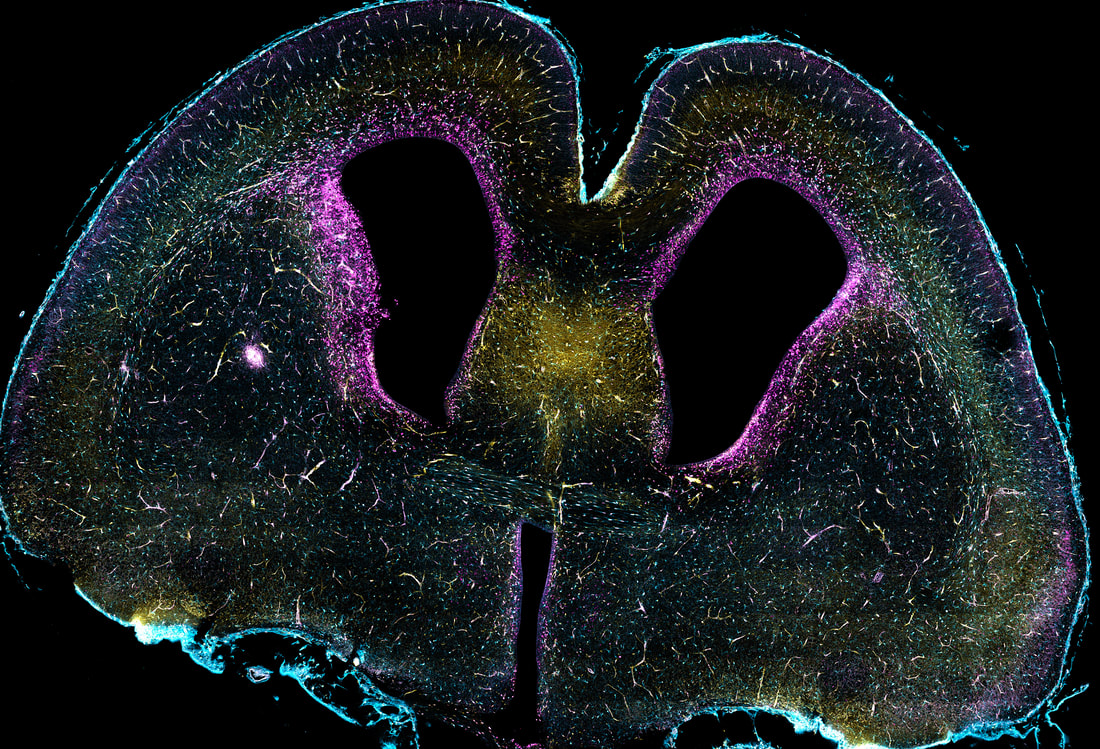
Throughout the CNS, neural progenitors first give rise to neurons before they start producing glia. The timing of this “neuron-glia switch” is determined by both cell-intrinsic factors and position-dependent extracellular cues. For example, the morphogen signaling molecule, Sonic hedgehog (Shh), is highly expressed in the embryonic ventral forebrain to pattern the dorsal-ventral axis. We discovered that Shh signaling increases in the dorsal forebrain at later developmental stages to initiate production of oligodendrocyte precursor cells (OPCs) from dorsal progenitors. This increase is facilitated by interneurons that migrate from ventral regions into the neocortex and secrete Shh ligands, thus providing a spatiotemporal mechanism for specifying the dorsal oligodendrocyte lineage.
Importantly, we found that the beginning of OPC production overlaps with the end of neurogenesis, so that only some progenitors form OPCs in response to Shh, while others continue to produce neurons. This suggests that additional pathways are required to specify subsets of progenitors that will initiate OPC production upon Shh exposure. To identify factors that might cooperate with Shh, we analyzed single cell RNA-sequencing datasets of the developing mouse neocortex, which revealed signatures of Notch pathway activation in Shh-responding progenitors. The Notch pathway is known to control cell fate specification, as perturbing Notch signaling early in development depletes progenitors and disrupts neuron production. Notch signaling is also important for OPC formation in the spinal cord, and we have recently shown that Notch signaling is required for normal OPC production in the dorsal forebrain.
We are now studying the interplay between the Notch and Shh signaling pathways and how they synergistically promote OPC formation. We currently testing the hypothesis that Notch primes pre-OPCs to respond to Shh signaling through its regulation of of the pioneer transcription factor, Ascl1.
Throughout the CNS, neural progenitors first give rise to neurons before they start producing glia. The timing of this “neuron-glia switch” is determined by both cell-intrinsic factors and position-dependent extracellular cues. For example, the morphogen signaling molecule, Sonic hedgehog (Shh), is highly expressed in the embryonic ventral forebrain to pattern the dorsal-ventral axis. We discovered that Shh signaling increases in the dorsal forebrain at later developmental stages to initiate production of oligodendrocyte precursor cells (OPCs) from dorsal progenitors. This increase is facilitated by interneurons that migrate from ventral regions into the neocortex and secrete Shh ligands, thus providing a spatiotemporal mechanism for specifying the dorsal oligodendrocyte lineage.
Importantly, we found that the beginning of OPC production overlaps with the end of neurogenesis, so that only some progenitors form OPCs in response to Shh, while others continue to produce neurons. This suggests that additional pathways are required to specify subsets of progenitors that will initiate OPC production upon Shh exposure. To identify factors that might cooperate with Shh, we analyzed single cell RNA-sequencing datasets of the developing mouse neocortex, which revealed signatures of Notch pathway activation in Shh-responding progenitors. The Notch pathway is known to control cell fate specification, as perturbing Notch signaling early in development depletes progenitors and disrupts neuron production. Notch signaling is also important for OPC formation in the spinal cord, and we have recently shown that Notch signaling is required for normal OPC production in the dorsal forebrain.
We are now studying the interplay between the Notch and Shh signaling pathways and how they synergistically promote OPC formation. We currently testing the hypothesis that Notch primes pre-OPCs to respond to Shh signaling through its regulation of of the pioneer transcription factor, Ascl1.
|
Developmental Biology | (2019) August 1;452(1):55-65.
Loss of Shh Signaling in the Neocortex Reveals Heterogeneous Cell Recovery Responses from Distinct Oligodendrocyte Populations Winkler CC, Franco SJ. Journal of Neuroscience | (2018) June 6;38(23):5237-5250. The Dorsal Wave of Neocortical Oligodendrogenesis Begins Embryonically and Requires Multiple Sources of Sonic Hedgehog Winkler CC, Yabut OR, Fregoso SP, Gomez HG, Dwyer BE, Pleasure SJ, Franco SJ. |